Fiberprint: Human Brain Wiring Shows Unique Fingerprint
The featured image was bought on Istock.com. Copyrights.
Abstract
White matter characterization studies use the information provided by diffusion magnetic resonance imaging (dMRI) to draw cross-population inferences. However, the structure, function, and white matter geometry vary across individuals. Here, we propose a subject fingerprint, called Fiberprint, to quantify the individual uniqueness in white matter geometry using fiber trajectories.
We learn a sparse coding representation for fiber trajectories by mapping them to a common space defined by a dictionary. A subject fingerprint is then generated by applying a pooling function for each bundle, thus providing a vector of bundle-wise features describing a particular subject’s white matter geometry. These features encode unique properties of fiber trajectories, such as their density along prominent bundles.
An analysis of data from 861 Human Connectome Project subjects reveals that a fingerprint based on approximately 3,000 fiber trajectories can uniquely identify exemplars from the same individual. We also use fingerprints for twin/sibling identification, our observations consistent with the twin data studies of white matter integrity.
Our results demonstrate that the proposed Fiberprint can effectively capture the variability in white matter fiber geometry across individuals, using a compact feature vector (dimension of 50), making this framework particularly attractive for handling large datasets.
Keywords: Subject fingerprint; dMRI; White matter geometry; Fiber trajectories; Sparse code pooling; HCP; Twin data
Characterizing the Brain of Individuals
Despite gross similarities, the brain of each individual shows unique characteristics in terms of structure and function. This uniqueness has been established by researchers using non-invasive imaging techniques like magnetic resonance imaging (MRI). Recently, there has been a growing interest in characterizing individual brains using a compact signature known as fingerprint [3,4,5]. This interest is mainly driven by improvements in data acquisition hardware and the increasing amount of publicly available data, thanks to large-scale initiatives like the Human Connectome Project [2] or the Human Brain Project. Brain fingerprinting could help understand the genetic basis of diseases like Parkinson’s, and is a significant step towards precision psychiatry [6].
Studies of the brain typically focus on its structure, function or connectivity. Structural studies investigate the various regions composing the brain, from the outer cortex to inner structures like the cerebellum or hippocampus, and their link to cognitive function or neurological diseases. In contrast, functional studies aim at identifying the brain regions of subjects that are activated at rest or when performing specific tasks. Finally, connectivity studies consider the geometry of brain fibers (i.e., myelinated axons) forming the brain’s white matter, which allow the different brain regions to communicate with one another. White matter connectivity is inferred using diffusion MRI (dMRI), a variant of MRI which observes the movement of water molecules along these fibers. While fingerprints have been proposed for brain structure and function, characterizing individuals using white matter connectivity has been elusive. This is due to various factors like variability across individuals, variable number and length of fiber trajectories inferred from dMRI.
Fiberprint: Identifying Individuals with the Wiring Architecture of their Brain
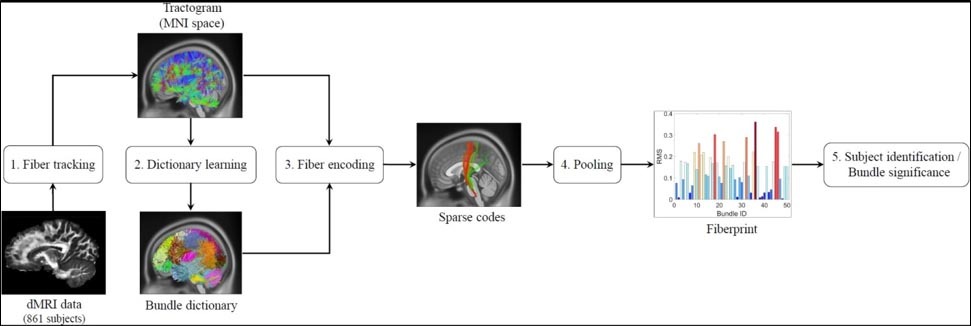
In this work, we propose a compact brain fingerprint, called Fiberprint, which captures the geometry of fiber trajectories, i.e., macroscopic inference of underlying fibers from dMRI (Figure 1).
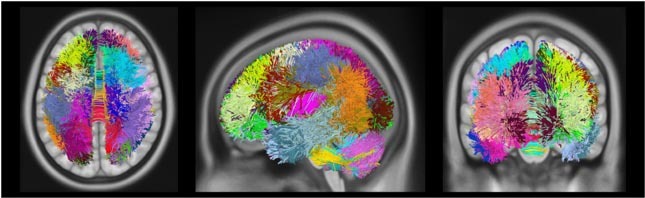
Figure 2 Dictionary visualization. Visualization of m=50 fiber trajectory prototypes learned from 10 subjects, with a unique color assigned to each dictionary prototype. For this simplified visualization each fiber is assigned to a single prototype.
The key idea is to build a common space representation for fiber trajectories, using concepts of sparse dictionary learning, and then to use this representation to encode a subject’s distribution of fiber trajectories into a fingerprint. This representation is modeled as a dictionary (Figure 2), the elements of which correspond to major fiber bundles in the brain.
This dictionary can be defined using an expert labeled set of fiber trajectories or an existing atlas. In this work, we learn it in an unsupervised manner using the data of ten unrelated subjects. The encoding of a fiber trajectory, known as sparse code, gives its membership to each of the dictionary bundles (Figure 3).
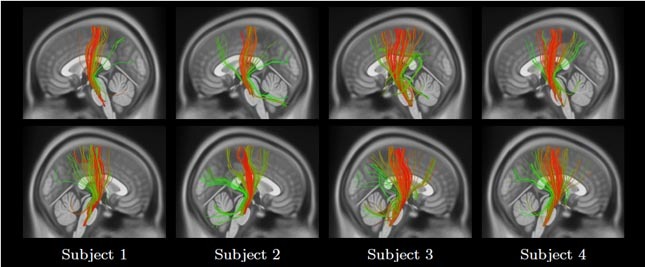
Figure 3 Color-coded visualization of fiber trajectory sparse code memberships for the left (top row) and right (bottom row) corticospinal bundles from four subjects. Green and red represent a low and a high membership of a fiber trajectory to a bundle.
This representation allows fiber trajectories to be assigned to multiple bundles, which is necessary when modeling overlapping bundles. The sparse codes of a subject’s fiber trajectories (Figure 4) are then aggregated into a single value for each bundle, estimating the density of fiber trajectories along this bundle.
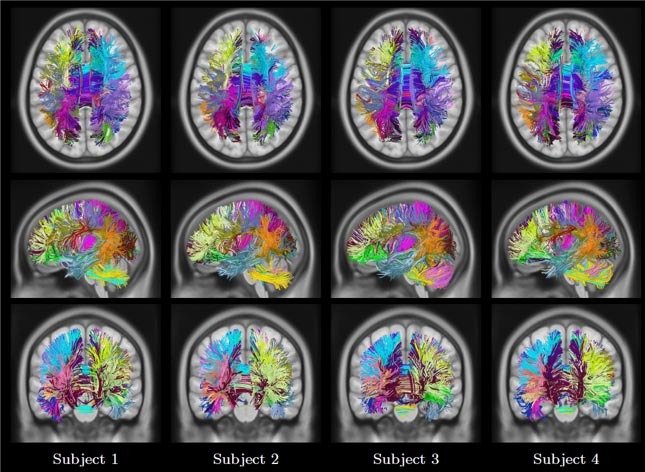
Figure 4 Visualization of fiber trajectory sparse code representation from four subjects. Each fiber trajectory is assigned to a single bundle by taking the maximum of the sparse code vector. Same color scheme as for Dictionary.
The proposed Fiberprint is obtained by stacking the pooling output for all dictionary bundles (Figure 5,6).
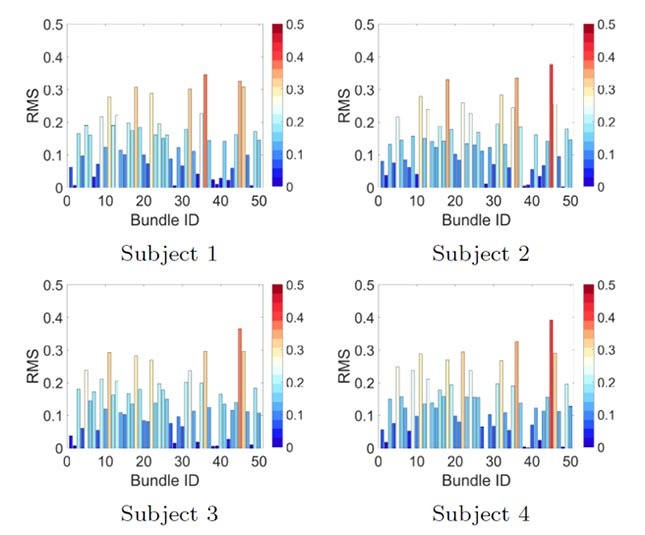
Figure 5 Subject fingerprint visualization. Color-coded bar plot representation for four subjects and RMS pooling functions, plotted as a value per bundle ID.
A large-scale analysis using the data of 851 subjects from the Human Connectome Project was conducted to measure our brain fingerprint’s ability to identify individual subjects, as well as their twin or non-twin siblings. This analysis revealed that a fingerprint of only 50 values, obtained with as little as 3,000 fiber trajectories, can uniquely identify a given person from the pool of subjects. The same fingerprint could also find a subject’s siblings with significant accuracy, demonstrating the link between genetic proximity and fiber geometry.
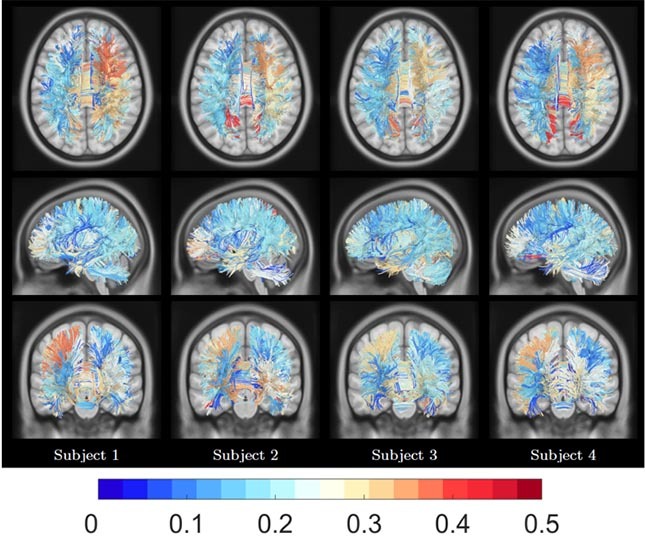
Figure 6 Subject fingerprint visualization. Color-coded bundles from four subjects representing the magnitude of their corresponding RMS pooling function values. Same color code scheme as for the bar plot representation
Future Research
One key aspect will be to study changes in the proposed fingerprint over time, in a longitudinal study. These changes could help understand how aging affects brain connectivity, from the perspective of fiber geometry. Similarly, fingerprint differences between healthy subjects and those suffering from a particular brain disease could reveal the involvement of fiber geometry in this disease.
Additional Information
To learn more on this research, please read the following article:
Kuldeep Kumar, Christian Desrosiers, Kaleem Siddiqi, Olivier Colliot, Matthew Toews, Fiberprint: A subject fingerprint based on sparse code pooling for white matter fiber analysis, NeuroImage, Volume 158, 2017, Pages 242-259, ISSN 1053-8119, http://dx.doi.org/10.1016/j.neuroimage.2017.06.083.