
Summary of Scientific Paper
Water treatment is often a complex and costly process for cities, involving the use of large quantities of chemicals and great resources to remove impurities from the water. To address this, chemically modified biodegradable fibres have been introduced into the current purification process to enhance their potential. Compared with conventional treatments—with a 45% contaminant removal—these fibres were able to remove up to 33% more contaminants (78%) and 60% more of certain types of microplastics than conventional treatments—80% vs. 20% respectively, for fibrous and conventional treatments. In conclusion, these fibres would make it possible to capture more contaminants currently resistant to conventional processes, and reduce the quantity of chemicals needed by the treatment industries. A recent study by Professor Lapointe—published in the prestigious Nature Water Journal—shows that our conventional plants are not efficient at removing nanoplastics, which are 1000 X smaller than microplastics [1].
Background
In the context of global population growth, preserving our drinking water resources, which represent only 2.8% of Earth’s total water content is crucial. Worldwide consumption of chemicals (coagulants) needed to purify water is 25 million tonnes per year, for a budget of $18 billion, leaving 7.8 million tonnes on average of toxic sludge, or waste, in the environment. [2] On the other hand, the facilities and surface area required for this treatment are equally important. For example, the Jean-R. Marcotte plant in Montreal is as large as 90 football stadiums [3], with an average daily treatment volume of 2.3 million cubic metres. This equals the scale of an Olympic stadium [4], requiring substantial use of construction materials and a budget of several million dollars for the purchase of chemicals.
To meet these challenges, it is vital to ensure continuous innovation in the way we purify water, both economically and ecologically. Our approach involves adding functionalized biodegradable fibrous materials to capture more contaminants in the water, making the purification process faster, more efficient, cleaner, less dependent on chemicals, and therefore less costly.
Water Treatment Through Coagulation, an Ongoing Improvement
One of the most popular methods of water purification involves adding certain chemicals to clump contaminants in the water, forming solid lumps known as flocs. This step is called coagulation. Using coagulation to purify water unfit for consumption is not a new concept.
This stage is followed by flocculation, which consists of helping the flocs formed during coagulation to clump together even further—increasing their size—and then allowing gravity to sink them to the bottom of a basin, leaving clean and clear water on the surface. The flocs that have settled to the bottom are then removed mechanically. This process is a common way of treating water, whether to make it drinkable in the plants or to remove impurities before releasing it back into nature.
Current Issues
Even though sophisticated automatic systems are used today, their main limitations are the time required for the flocs to sink to the bottom of the settling tanks and the space and construction materials needed to build these large tanks. In addition, the emergence of relatively recent and difficult-to-capture contaminants from industry and domestic use, such as microplastics, further complicates the operation and final choice of processes used in the plants. Finally, chemical product purchasing costs are also an issue.
Fibre Innovation
The innovation in our research work consists of adding biodegradable fibres to the same process, chemically modified to capture more contaminants (by adsorption), notably from recycled paper and mining residues, reinforcing the ability of coagulants and flocculants to cluster more impurities in the water. Instead of forming small flocs, these fibres help create larger, more efficient flocs, facilitating the separation and removal of contaminants from the water during decantation—a process used worldwide. They can be compared to very powerful magnets that stick contaminants together. Their mode of operation is relatively simple: the fibres are injected in certain doses and at very specific times during the purification process in order to reach their maximum potential. They significantly improve water treatment efficiency by increasing the size of impurity clusters—10 to 100 times larger—see Figure 1.
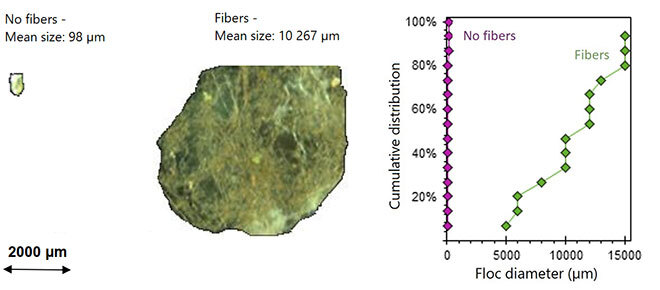
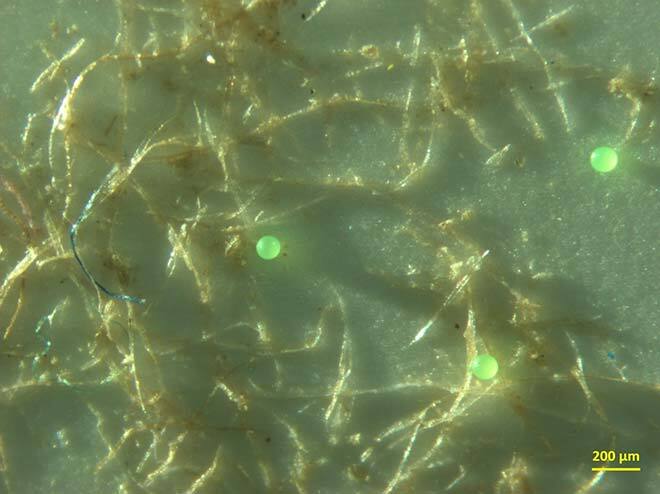
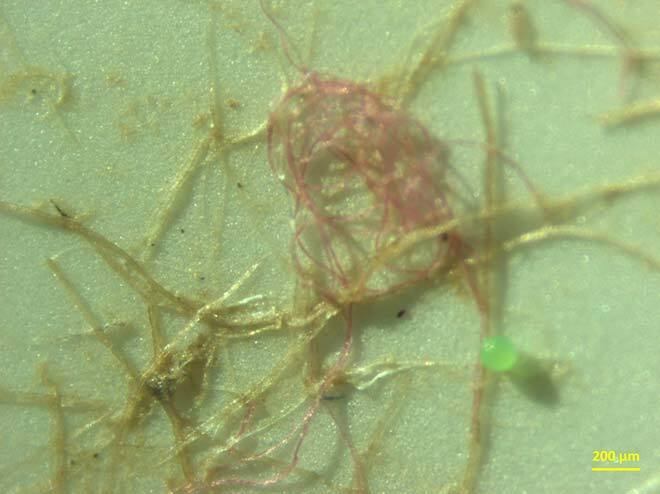
What is more, these fibres can capture certain types of microplastics and textile fibres (see Figure 2) at a 20% to 60% greater efficiency than the normal process used by many large cities [5]. Last but not least, these fibres make it possible to reduce the size of the tanks needed for floc decantation, making it possible to replace them with modern sieving technology that filters water to achieve better results over a smaller surface area. This makes the process more economical and environmentally friendly.
Conclusion
This innovation makes it possible to reduce or even eliminate the decantation stage by replacing the decanting tank with a rotary screen. This saves construction materials and reduces chemical costs for cities. It also captures contaminants that were previously very difficult to remove, ensuring cleaner, microplastic-free water that can be reused for irrigation in agriculture. The aim is to prevent these contaminants from ending up on consumers’ plates.
Additional Information
For more information on this research, please see the following paper:
Walid Alioua, Mathieu Lapointe, Fiber-based super-bridging agents enable efficient contaminant removal via settling and screening: Impact on microplastics, textile fibers, and turbidity, Journal of Environmental Chemical Engineering, Volume 11, Issue 3, 2023, 110195,
ISSN 2213-3437.
Sources :
[1] https://www.nature.com/articles/s44221-023-00177-3
[2] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7126837/
[4] https://montreal.ca/articles/desinfection-lozone-travaux-la-station-depuration-jean-r-marcotte-27451
[5] https://www.sciencedirect.com/science/article/abs/pii/S221334372300934X



